
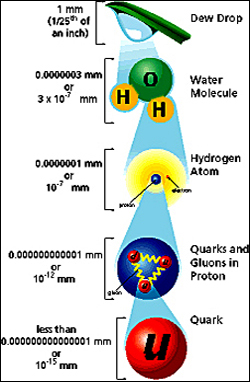
A Brief Particle History
Atoms, Electrons And Protons
English chemist, physicist and meteorologist John Dalton is considered the father of modern atomic theory. In 1805 Dalton published a paper which included the following main points of atomic theory:
- Elements are made of extremely small particles called atoms.
- Atoms of a given element are identical in size, mass, and other properties.
- Atoms of different elements differ in size, mass, and other properties.
- Atoms cannot be sub-divided. (of course we can do this today).
- Atoms of different elements combine in simple whole-number ratios to form chemical compounds.
- In chemical reactions, atoms are combined, separated, or rearranged.
(John Dalton was also famous as a meteorologist and the cooling period from 1790 to 1830 is named after him - The Dalton Minimum. This cooling period is believed to be due to lower than normal sun activity which was indicated by smaller sunspot cycles.)
The physicist J. J. Thomson, through his work on cathode rays in 1897, discovered the electron and concluded that they were a component of every atom. Thomson suggested that the very small mass, negatively charged electrons were distributed throughout the atom. He explained that their negative charge was balanced by the presence of a uniform sea of positive charge. He also hypothesized that they might rotate in rings.
In 1909, physicist Ernest Rutherford and associates bombarded a sheet of gold foil with alpha rays, by then known to be positively charged helium atoms. They discovered that a small percentage of these particles were deflected through much larger angles than predicted. Rutherford interpreted the results of the experiment as the positive charge of the gold atom and most of its mass were concentrated in a nucleus at the center of the atom.
In 1913, physicist Niels Bohr demonstrated that electrons were confined into clearly defined, quantized orbits, and could jump between these orbits, but could not spiral inward or outward in intermediate states. An electron must absorb or emit specific amounts of energy to transition between these fixed orbits. In the same year Ernest Rutherford and Antonius Van den Broek presented a model which proposed that each atom contains a number of positive nuclear charges in its nucleus that is equal to its atomic number in the chemistry Periodic Table. Top
Quantum Theory

In 1924, Louis de Broglie suggested that particles behave like waves. In 1926, Erwin Schrödinger used this idea to develop a mathematical model of the atom that described electrons as three dimensional waveforms rather than "point" particles. This was the beginning of Quantum Mechanics (or Quantum Theory). A consequence of using waveforms to describe particles is that it is impossible to obtain precise values for both the position and velocity of a particle at the same time. This became known as the "uncertainty principle" formulated by Werner Heisenberg in 1926. This model was able to explain observations of atomic behavior that previous models could not. Afterwards the planetary model of the atom was discarded in favor of one that described atomic orbital zones around the nucleus where a given electron is most likely to be observed. See the sketch at the left.
In 1932 the neutron, a neutral-charged particle with a mass similar to the proton, was discovered by physicist James Chadwick in Cambridge, England who received the Nobel prize for his work. Isotopes of elements were then explained as having the exact same number of "protons" as the original element, but a different number of "neutrons" within the nucleus.
In 1938, the German scientist Otto Hahn, conducting experiments on radioactivity, directed neutrons into uranium atoms which resulted in the first nuclear fission reaction. In November of 1945 the Swedish Academy awarded Hahn the Nobel Prize in Chemistry retroactive to 1944. Near the end of World War II in early 1945 Hahn had given himself up to British authorities and was being interned in England at the time. Otto Hahn, regarded as "the father" of nuclear chemistry, had nothing to do with the German war efforts and had vigorously opposed the persecution of Jews. Top
Quarks And Leptons

In the 1950s, the development of improved particle accelerators and particle detectors allowed scientists to study collisions of atoms moving at very high energies. In 1958 scientists at the Stanford Linear Accelerator Center (SLAC) found that neutrons and protons contained more fundamental particles - quarks. Quarks were named by Murray Gell-Mann of Caltech, who won the Nobel Prize in 1969 for his work on them. Neutrons and protons now fall into the family of particles called "hadrons". which are particles composed of the smaller more elementary "quarks". Hadrons are made of quarks and form two other families: baryons, normal stable matter made of three quarks, and mesons, short lived unstable particles made of one quark and one anti-quark. See the chart at the left. Protons and neutrons are baryons. Different combinations of protons and neutrons make up the nuclei of atoms. The complete atom also contains electrons, which are leptons, in addition to the nucleus. Finally, different combinations of atoms make up molecules.
Throughout the 1950's and 1960's, a bewildering variety of particles were found from scattering experiments using advanced particle accelerators. At the time, this assortment of particles was referred to as the "particle zoo" and a new particle was found just about once a month. During the 1970's, a large number of these particles were found to be combinations of a relatively small number of fundamental particles. Models were then developed that successfully explained the properties of atomic nuclei in terms of the fundamental particles and the forces that govern their interactions. These observations and theories have been thoroughly tested and combined into what is now known as the Standard Model. Physicists have identified 12 elementary particles called Fermions that are the fundamental elements of "matter". At this time it is believed that these 12 elementary particles cannot be further sub-divided. Top
Fermions - Generation I

Our everyday world is made of just "three" of the Fermion building blocks: the up quark, the down quark and the electron. These are the "stable" primary elements and these three particles are all that's needed to form atoms and and then much larger molecules. The electron neutrino, observed in the decay of other particles, completes the "First Generation" of three building block families of Fermions. See the Fermion chart at the left.
A proton consists of two up-quarks and one down-quark. A neutron consists of one up-quark and two down-quarks. Neutrinos, predicted in the 1930's, were found in the mid 1950's. Neutrinos were very difficult to find because they rarely interact with other matter. Billions of neutrinos, ejected by the sun, are passing through our bodies and the earth right now. The electron and electron neutrino are called "leptons". The characteristic that separates quarks from leptons is a property called "color charge". This has absolutely nothing to do with color in its usual sense. (Quarks are much smaller than the wavelength of visible light so they do not have any color per se.) The "color charge" indicates whether or not the particle interacts with the "strong nuclear force". See the Color Charge section below. Quarks "do" interact with the strong nuclear force and leptons "do not". More on the "strong nuclear force" on the Force Page.
All the fundamental particles exhibit some form of an "electric charge". The electric charge is described in fractions of the "proton charge" (i.e. 0, 1/3, 2/3, 1). The electron group has a charge of -1. The hydrogen atom has a proton with a +1 charge and an electron with a -1 charge and hence hydrogen is electrically neutral and very stable. The neutrino group all have 0 charge, while the quarks have +2/3 or -1/3 charge of the proton. A neutron is made of one up quark ( a charge of +2/3) and two down quarks (2 times -1/3 = -2/3) so it too is electrically neutral.
For every particle, physicists have discovered a corresponding anti-particle, which looks and behaves almost the same way. Anti-particles, though, have the opposite properties of their corresponding particles. An anti-proton, for example, has a negative electric charge while a proton is positively charged. As soon as an anti-particle and a particle meet, they annihilate, disappearing in a flash of energy. However, anti-matter is as real as normal matter, its the name that is somewhat confusing. Top
Fermions - Generations II and III
Muons, 200 times heavier than electrons, were discovered by Carl D. Anderson and Seth Neddermeyer at Caltech in 1936 while studying cosmic rays from from outer space. Anderson noticed that some particles curved differently from electrons when passing through a magnetic field. They were negatively charged but curved less sharply than electrons. The muon is an elementary particle similar to the electron, with an electric charge of -1, and a spin of ½ (see next section on spin). The muon is an unstable subatomic particle with a mean lifetime of 2.2 microseconds. Muons quickly decay into three other particles, an electron and two different types of neutrinos.
The muon was the first particle discovered in the Generation II family of particles. Using ever more powerful accelerators, scientists discovered more and more fundamental particles in the debris from particle collisions. Physicists recognized a pattern among the particles and organized them into the three families we now call Generations I, II, and III. See the three generation chart of matter above. Each family consists of two quarks, a neutrino and an electron or one of its cousins. The particles in Generations II and III are much heavier than Generation I particles and are very unstable. Generation II and III particles last just microseconds before they decay into the particles of Generation I. The exception are neutrinos which do not decay at all. Top
Mysterious Neutrinos

All three neutrinos are leptons, the family of particles that includes electrons. Neutrinos do not feel the strong force that holds together protons and neutrons in an atomic nucleus. Because they do not have any electrical charge, neutrinos also do not feel the electromagnetic force. Almost all neutrinos pass right through the earth without ever interacting with an earthly atom. Only the force of gravity and the weak nuclear force (the force responsible for radioactive decay) interact with the three known neutrino versions.
Neutrinos were first predicted by Wolfgang Pauli in 1930, who said "I have postulated a particle that can not be detected". Neutrinos were not detected until 1956 by Clyde Cowan et. al. who were awarded the Nobel Prize in 1995 (yes 1995, 49 years later). Subterranean laboratories are ideal for neutrino detectors. Thick layers of rock are perfect shields to block other types of particles that can not penetrate deeply. Most neutrino facilities are in old converted mines. Buried deep underground, in the inactive Soudan Iron Mine in Minnesota, is the 5,400 metric ton octagonal MINOS detector shown at the upper left in the photo above. (To the right of the detector is a neutrino inspired mural by artist Joseph Giannetti. For more neutrino information visit the Neutrino Page. Top
Spin
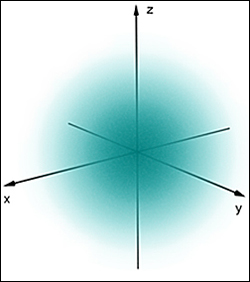
All elementary articles have an intrinsic property called "spin" which is not easy to visualize. Conceptually spin is similar to a planet spinning on its axis. As the name suggests, spin was originally conceived as the rapid rotation of a particle around an axis. However as scientists eventually learned, elementary particles do not have any axis per se. For example, an electron is not a particle in space that rotates and orbits about a nucleus. They are more like 3 dimensional standing waves in a cloud. They do exhibit a tiny magnetic field as if they rotated extremely rapidly, independent of their orbital rotation around the nucleus. See the "electron cloud" probability distribution to the left. Their specific location can not ever be known, only the probability of being in any given location is known - the uncertainty principle.
In our "normal" world, charged objects that spin have certain magnetic properties. In the micro world, elementary particles exhibit the same magnetic properties, hence the spin analogy. However, spin is only an analogy and should not be taken rigorously. The spin connection is also correct in so far as spin obeys the mathematical laws of angular momentum. Although the direction of a particle's spin can be changed, the particle cannot be made to spin faster or slower. All elements of a particular group have the exact same spin, it is part of their intrinsic makeup. Furthermore, spin is quantized - meaning that only certain discrete spins are allowed. For example, all Fermions have a spin of 1/2. Many physical processes, ranging from the smallest nuclear dimensions to the largest astro-physical distances, depend greatly on interactions of subatomic particles and the spin of those particles.
All the known particles in the universe can be divided into two groups: particles with a spin of 1/2 - all the matter (Fermions) in the universe, and particles with a spin of 0, 1, and 2 - all the forces (bosons) that interact with the matter particles. (For more info on forces, see the Force Page.) All the matter particles (spin 1/2) obey the Pauli exclusion principle. This was discovered in 1925 by Austrian physicist Wolfgang Pauli, who received the Nobel Prize in 1945 for his work. The Pauli exclusion principle states that no two Fermions (matter particles) can occupy the same state, i.e. they both can not have the same position and velocity. It is the Pauli exclusion principle that separates all the elements into different categories that make up the Periodic Table in chemistry. Top
Color Charge
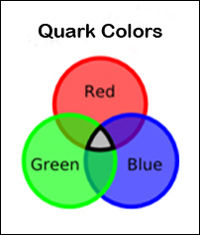
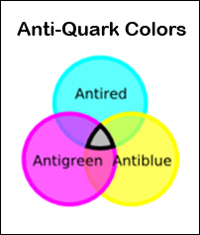
The "color charge", found only in quarks and gluons, is completely unrelated to human perception of color. The color charge is an inherent property of quarks and gluons that is relative to the "strong nuclear force". The term "color" was chosen because the property to which it refers has three aspects. They are analogous to the three primary colors of red, green, and blue, which when combined yield white. A quark's color can take one of three values, also called red, green, and blue. An anti-quark can take one of three anti-colors, called anti-red, anti-green, and anti-blue (represented as cyan, magenta and yellow). Gluons exhibit two colors, such as red and anti-green, which constitute their color charge. Color charge has a very small range, only the size of an atomic nucleus.
Color confinement is a phenomenon that "color charged" particles (quarks and gluons) exhibit. It means they cannot be isolated or be directly observed individually. Quarks and anti-quarks come in groups of two or three to form "hadrons". The two types of hadrons are baryons (three quarks) and mesons (one quark and one anti-quark). Individual quarks are never found alone and cannot be separated from their parent hadron. This is why quarks can only be observed at the hadron level.
The "color charge" is similar to the electromagnetic charge, except it comes in three types instead of two, and it is a different type of force with different rules of behavior. (These rules are explained by the theory of quantum chromodynamics (QCD), which is the theory of quark-gluon interactions.) Similar to the additive color model in basic optics, the combination of three quarks or three antiquarks, each with different "color charges", will always result in a total "white" color charge. All hadrons have "zero total" color charge, i.e. "white".
The "strong nuclear force" between quarks is mediated (enabled) by gluons. As mentioned above, each gluon carries one color charge and one anti-color charge. Since gluons carry color charge, they themselves are able to emit and absorb other gluons. Gluons are constantly exchanged between quarks. When a gluon is transferred between two quarks, a color "change" occurs in the two quarks and also in the gluon. While each quark's color is constantly changing, the hadron's total color charge remains "zero" (or white) and the "strong nuclear force" is conserved. Top
The Higgs Particle
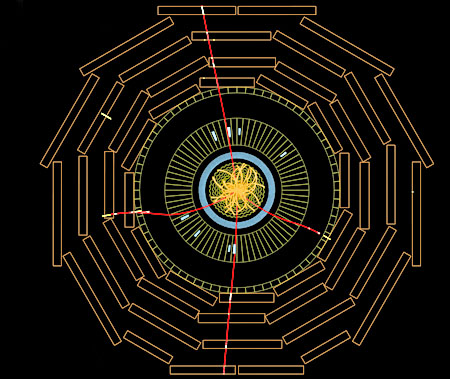
The new particle announced in July, 2012 was the Higgs boson, the particle whose field explains how other particles get their mass. CERN physicists had announced in early 2012 with 99% certainty that they had found a new elementary particle weighing about 126 times the mass of a proton. After collecting two and a half times more data, the physicists said the particle was definitely a Higgs boson. The CMS image to the left is believed to be a collision resulting in a Higgs particle temporarily being formed and then almost instantly decaying into four electrons.
"To me it is clear that we are dealing with a Higgs boson though we still have a long way to go to know what kind of Higgs boson it is," said CMS spokesperson Joe Incandela. The scientists are not sure whether this particular Higgs boson is the one predicted by the Standard Model or perhaps the lightest of several bosons predicted to exist by other theories. The Higgs particle should have zero spin, its parity should be positive, and its mass should be between 120 and 130 GeV. All of these attributes were confirmed by data from both the ATLAS and CMS experiments.
Observing how this particular particle decays into other particles will let physicists know whether this Higgs particle is the "plain vanilla" Standard Model Higgs. Detecting a Higgs boson is rare occurrence. Just one is observed for every one trillion proton to proton collisions. Therefore, the CERN physicists say they need much more data to understand all of the ways in which the Higgs decays. This will not occur until 2015 or later when the Large Hadron Collider (LHC) starts running again and more data at higher collision speeds is collected by the ATLAS and CMS teams. For more information on the Higgs particle, see Hunting The Higgs Particle. Top
Could A Bubble Burst Our Universe?

The Cosmological Standard Model, the current theory of the universe, has equations that depend in part on the mass values of both the "Higgs boson" and the "top quark". The top quark's mass is estimated at 171 GeV and the Higgs boson is estimated at 126 GeV. When these values are plugged into the equations, the theoretical universe turns out to be only temporarily stable and becomes "unstable" over a time frame of billions of years. In this context, what does "unstable" mean?
According to quantum theory, the lowest energy state of our current universe is not the lowest possible state. In this theory, there exists an even lower energy state, one that our universe would most likely transition to if disrupted. That might not sound too ominous until it is understood that in this new lower energy state, all the protons in all the matter in the universe will decay at the speed of light into more elementary particles. The unfortunate side effect of this new state is that all the stars, the planets, the sun, the earth, and us humans would no longer exist.
If a phase transition of this kind were to happen, a bubble of the new phase would suddenly appear within the old phase (our universe) and expand close to light speed. See the artist illustration at the left. The new bubble would very rapidly grow and over time devour the current universe, converting everything to the new phase. It would be the end of the universe as we know it today.
This doomsday idea has received quite a bit of publicity in the scientific media. So lets consider the likelihood of this event coming to fruition (as opposed to making a media splash). The furore in the media has resulted in a re-examination of the the Cosmological Standard Model equations. The result is that for the universe to be stable, the Higgs mass needs to be larger than 129.4 ± 5.6 GeV (123.8 to 135.0 GeV). Therefore at 126 GeV, the Higgs mass does fit within the statistical limits for the universe to be stable. In addition, almost all physicists believe the Standard Model is very incomplete. For example, it does not include gravity, or the experimentally observed mass of neutrinos, or dark matter, or dark energy. So most physicists believe the Standard Model will undergo serious revisions in the future and its implications will most certainly be revised.
Even if a destructive bubble were to originate somewhere in the universe and travel at the speed of light, our universe is many billions of light years across. Remember, the "observable" universe is roughly 90 billion light years wide and the "total universe" is believed to be at least 1000 times as wide as the observable universe. So if a destructive bubble were to suddenly appear somewhere and travel in all directions at light speed, it would likely take many billions of years to reach us. Keep in mind that our sun, and the earth, will self destruct in about 5 billion years.
Finally, there are indications from the data collected at the Large Hadron Collider at CERN that it is very unlikely that we live in an unstable universe. This implies that humanity is safe for a very, very long time, most likely as long as the sun and earth survive.